Generalized pustular psoriasis (GPP) is a serious, neutrophilic, chronic and persistent autoinflammatory condition.
The clinical course of GPP is unpredictable and highly heterogeneous. GPP flare frequency, severity, and duration vary between patients and between flares. Significant GPP symptoms are associated with flares, including itching, burning, pain, erythema, sterile pustules and scaling, affecting patients’ ability to perform everyday activities. Importantly, a flare can be potentially life-threatening which is why a GPP diagnosis should not be delayed by any predefined duration of pustulation or extent of body surface area.
Patients need a treatment that addresses the acute and chronic phases of GPP. The National Psoriasis Foundation strongly advocates for timely access to FDA-approved treatments for GPP.
So, what is SPEVIGO? SPEVIGO is the only FDA-approved treatment for GPP.
GPP is caused by dysregulated IL-36 receptor signaling, leading to a neutrophilic inflammatory response.
SPEVIGO is the only treatment that inhibits signaling in the IL-36 pathway, the key inflammatory pathway in GPP.
The binding of SPEVIGO to the IL-36 receptor prevents the subsequent activation of IL-36 receptor and as a result, downstream activation of pro-inflammatory and profibrotic pathways does not occur. The precise mechanism linking reduced IL-36 receptor activity and the treatment of GPP flares is unclear.
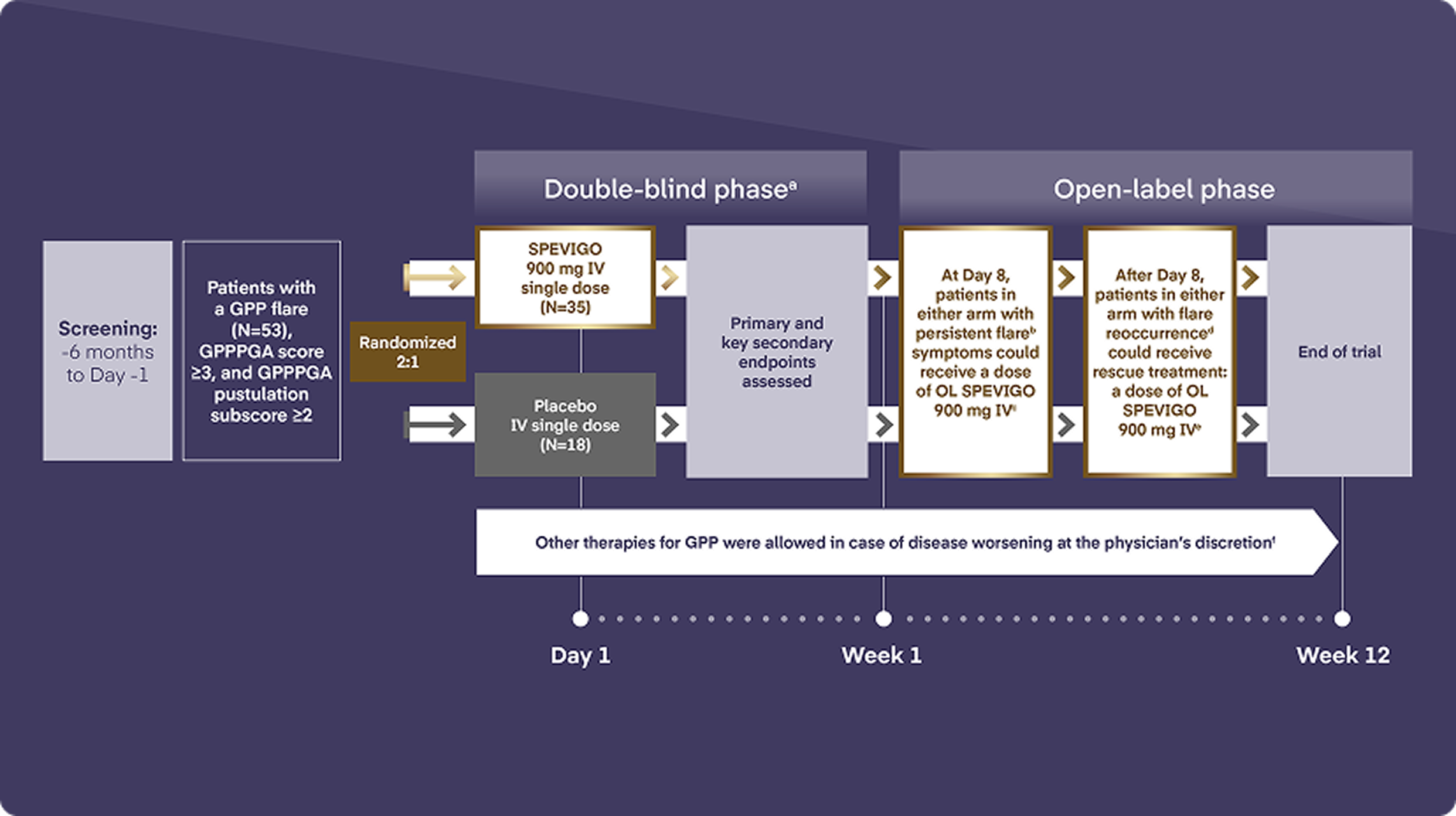
Let’s take a look at the EFFISAYIL 1 trial, which investigated the efficacy and safety of SPEVIGO intravenous infusion compared to placebo in patients experiencing a GPP flare. To be included, patients had to be experiencing a GPP flare of moderate to severe intensity that affected at least 5% of the body surface area with erythema and the presence of pustules. Patients also had to have evidence or prior evidence of systemic symptoms.
Patients were randomly assigned to receive a single 900 mg intravenous dose of SPEVIGO or placebo.
Patients experiencing persistent symptoms at Day 8 or a new flare after Day 8 until the end of the trial were eligible to receive open-label 900 mg SPEVIGO IV.
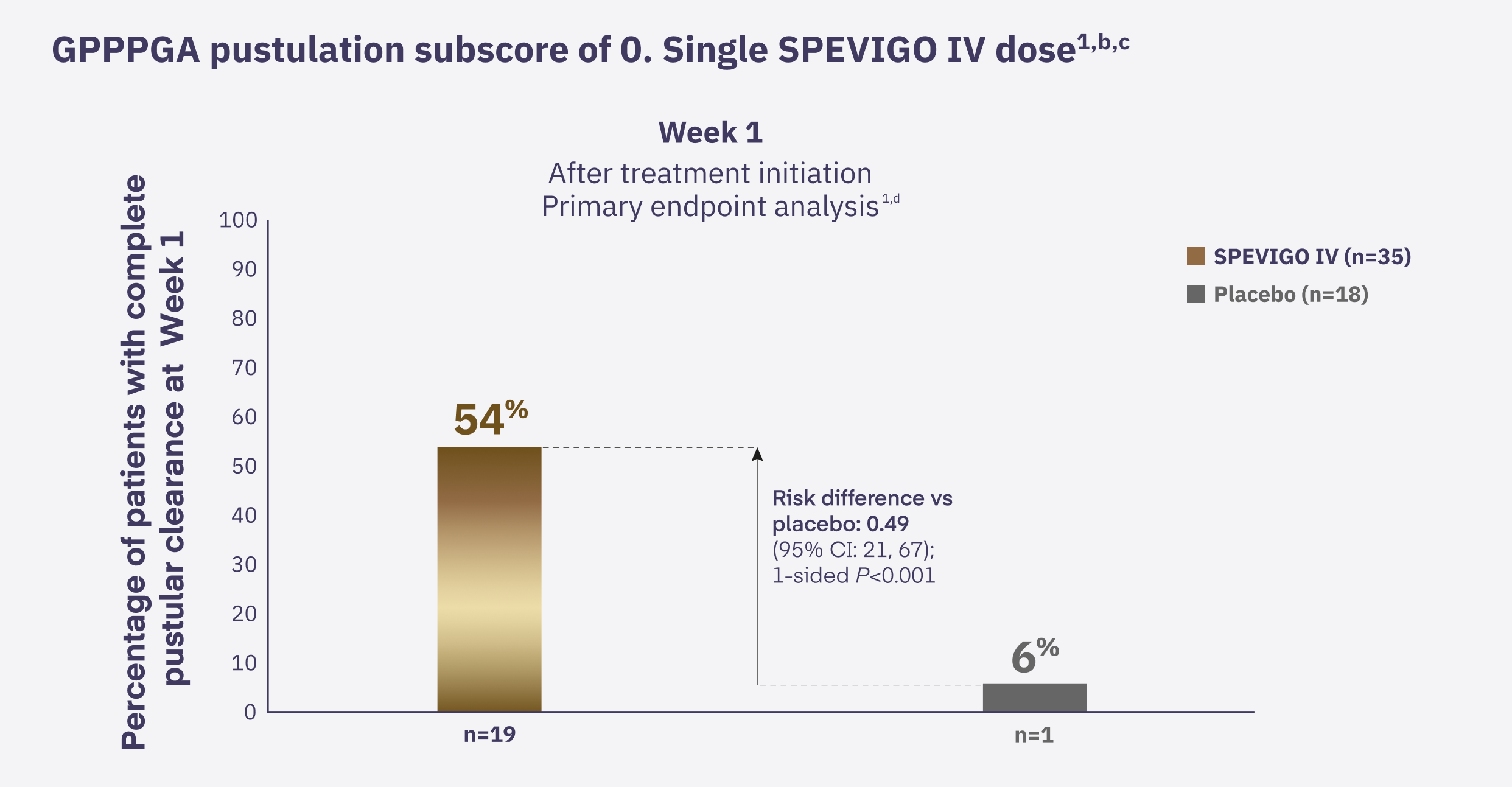
In EFFISAYIL 1, 54% of patients achieved rapid pustular clearance with SPEVIGO IV at Week 1.
For instance, a patient in EFFISAYIL 1 who had a GPP pustulation subscore of 3 at baseline, after a single dose of SPEVIGO IV, achieved rapid pustular clearance at Week 1. The patient continued to remain flare-free at Week 4.
During EFFISAYIL 1, asthenia and fatigue, headaches and nausea, were the most commonly observed adverse reactions with SPEVIGO IV for the treatment of GPP flares that occurred more frequently than the placebo group through Week 1.
In EFFISAYIL 1, additional adverse reactions that occurred through week 12 in subjects treated with 1 single dose of randomized SPEVIGO were mild to moderate infections, including device-related infection, subcutaneous abscess, furuncle and influenza. Please see additional important safety information on this website and the full prescribing information for SPEVIGO.
In conclusion, in patients with a GPP flare that affected at least 5% of the body surface area with erythema and pustules, 54% of patients achieved rapid pustular clearance with SPEVIGO IV at Week 1. The most common adverse reactions are asthenia and fatigue, headache, nausea, pruritus and prurigo, infusion site hematoma and bruising, and urinary tract infection. SPEVIGO is the only FDA-approved treatment for GPP.
Indication
SPEVIGO is indicated for the treatment of generalized pustular psoriasis (GPP) in adults and pediatric patients 12 years of age and older and weighing at least 40 kg (88 lb).
Important Safety Information
CONTRAINDICATIONS
SPEVIGO is contraindicated in patients with severe or life-threatening hypersensitivity to spesolimab-sbzo or to any of the excipients in SPEVIGO. Reported hypersensitivity reactions have included drug reaction with eosinophilia and systemic symptoms (DRESS) and anaphylaxis.
WARNINGS AND PRECAUTIONS
Infections: SPEVIGO may increase the risk of infections. In patients with a chronic infection or a history of recurrent infection, consider the potential risks and expected clinical benefits of treatment prior to prescribing SPEVIGO. Treatment with SPEVIGO is not recommended in patients with any clinically important active infection until the infection resolves or is adequately treated. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur during or after treatment with SPEVIGO. If a patient develops a clinically important active infection, discontinue SPEVIGO therapy until the infection resolves or is adequately treated.
Risk of Tuberculosis: Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with SPEVIGO. Avoid use of SPEVIGO in patients with active TB infection. Consider initiating anti-TB therapy prior to initiating SPEVIGO in patients with latent TB or a history of TB in whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during and after SPEVIGO treatment.
Hypersensitivity and Infusion-Related Reactions:
- Serious hypersensitivity reactions, including anaphylaxis and delayed reactions such as drug reaction with eosinophilia and systemic symptoms (DRESS), have been reported during and following administration of SPEVIGO. These reactions can occur with the first dose or subsequent doses.
- SPEVIGO is contraindicated in patients with severe or life-threatening hypersensitivity to spesolimab-sbzo or to any of the excipients in SPEVIGO. If a patient develops signs of anaphylaxis or other serious hypersensitivity, discontinue SPEVIGO immediately and initiate appropriate treatment.
- If a patient develops mild or moderate hypersensitivity during an intravenous infusion or other infusion-related reactions, stop SPEVIGO infusion and consider appropriate medical therapy (eg, systemic antihistamines and/or corticosteroids). Upon resolution of the reaction, the infusion may be restarted at a slower infusion rate with gradual increase to complete the infusion.
Vaccinations: Prior to initiating SPEVIGO for treatment of GPP, complete all age-appropriate vaccinations according to current immunization guidelines. Avoid use of live vaccines in patients during and for at least 16 weeks after treatment with SPEVIGO. No specific studies have been conducted in SPEVIGO-treated patients who have recently received live viral or live bacterial vaccines.
ADVERSE REACTIONS
Intravenous SPEVIGO for Treatment of GPP Flare (Study Effisayil-1): Most common adverse reactions reported in ≥5% of patients treated with SPEVIGO in the clinical trial were asthenia and fatigue, headache, nausea, pruritus and prurigo, infusion site hematoma and bruising, and urinary tract infection (UTI).
Specific Adverse Reactions
- Infections: The most frequent adverse reactions that occurred in subjects treated with intravenous SPEVIGO were infections. During the 1-week placebo-controlled period in Study Effisayil-1, infections were reported in 14% of subjects treated with SPEVIGO compared with 6% of subjects treated with placebo. Serious infection (UTI) was reported in 1 subject (3%) in the SPEVIGO group and no subjects in the placebo group. Infections observed through Week 1 in Study Effisayil-1 in subjects treated with SPEVIGO were mild (29%) to moderate (71%).
- Drug Reaction With Eosinophilia and Systemic Symptoms (DRESS): Two cases of DRESS were reported in Study Effisayil-1 in subjects with GPP who were treated with intravenous SPEVIGO. RegiSCAR DRESS validation scoring (with the following categories: “no,” “possible,” “probable,” or “definite” DRESS) was applied to the reported cases. Reported cases were assessed as “no DRESS” and “possible DRESS.”
Subcutaneous SPEVIGO for Treatment of GPP When Not Experiencing a Flare (Study Effisayil-2): Regarding the exposure-adjusted incidence rates for subjects on randomized treatment prior to receiving rescue treatment for flare or completing trial without a flare, the rate per 100-patient years for injection site reaction (including erythema, pain, swelling, induration, urticaria, and warmth at the injection site) was 31.6 for the subcutaneous SPEVIGO cohort (600 mg loading dose followed by 300 mg every 4 weeks) vs 12.7 for the placebo cohort. The rate per 100-patient years for UTI was 18 for SPEVIGO vs 0 for placebo. The rate per 100-patient years for pruritus was 8.8 for SPEVIGO vs 0 for placebo. The rate per 100-patient years for arthralgia was 13.3 for SPEVIGO vs 6 for the placebo cohort. There were 3 subjects who discontinued subcutaneous SPEVIGO due to treatment-emergent adverse events of psoriasis compared to no subjects in the placebo cohort who discontinued placebo for any treatment-emergent adverse event.
Safety in Study Effisayil-2 After Flare: In Effisayil-2, subjects who experienced a GPP flare and received at least one dose of an open-label single intravenous 900 mg dose of SPEVIGO were treated with open-label subcutaneous SPEVIGO 300 mg. These subjects (n=19) received subcutaneous dosing at every 12 weeks, which could have been increased to every 4 weeks based on GPPPGA total score or pustulation subscore increased by ≥1 from any previous open-label maintenance visit. The reported safety profile of open-label subcutaneous SPEVIGO use after treatment of GPP flare with open-label intravenous SPEVIGO use was consistent with the safety profiles of use of SPEVIGO from Trial Effisayil-1 and randomized controlled data from Trial Effisayil-2.
Clinical Development of Spesolimab-sbzo
- Guillain-Barre Syndrome (GBS): Among approximately 835 subjects exposed to spesolimab-sbzo during clinical development, GBS was reported in 3 subjects who received various doses of spesolimab-sbzo via various methods of administration in clinical trials for unapproved indications.
SPECIFIC POPULATIONS
Pediatric Use: The safety and effectiveness of SPEVIGO for the treatment of GPP have been established in pediatric patients 12 years of age and older and weighing at least 40 kg. Use of SPEVIGO for this indication is supported by data from a randomized, placebo-controlled study, which included 6 pediatric subjects 14 to 17 years of age with a history of GPP treated with subcutaneous SPEVIGO (Study Effisayil-2), and evidence from an adequate and well-controlled study of intravenous SPEVIGO in adults with GPP (Study Effisayil-1), with additional pharmacokinetic analyses showing similar drug exposure levels in adults and pediatric subjects 12 years of age and older and weighing 40 kg or more. The safety and effectiveness of SPEVIGO in pediatric patients younger than 12 years of age or in pediatric patients weighing less than 40 kg have not been established.
Please see SPEVIGO Prescribing Information, including Medication Guide.